RNA-based medicines are a class of treatments that leverage RNA molecules to target and modulate...
Lipid Nanoparticles in RNA therapeutics
RNA therapeutics represent a groundbreaking approach in modern medicine, leveraging RNA molecules to modulate gene expression, silence disease-causing genes, or introduce therapeutic proteins.
Unlike DNA therapies, which require nuclear entry and risk genomic integration, RNA treatments act in the cytoplasm, allowing precise, transient gene modulation with lower long-term risks.
However, widespread adoption has been limited by RNA’s instability, rapid degradation by nucleases, and poor cellular uptake due to its negative charge and hydrophilic nature. These challenges reduce bioavailability and therapeutic efficacy in vivo.
Delivery of RNA-based medicines with lipid nanoparticles (LNPs)
Lipid Nanoparticles (LNPs) are nanoscale delivery systems composed of lipid-based structures that encapsulate and protect small molecules and acid nucleics.
They are essential in RNA therapeutics, shielding RNA from degradation, enhancing cellular uptake, and enabling endosomal escape to reach target sites. By overcoming key delivery barriers, LNPs have driven breakthroughs in mRNA vaccines, gene editing, and treatments for genetic disorders.
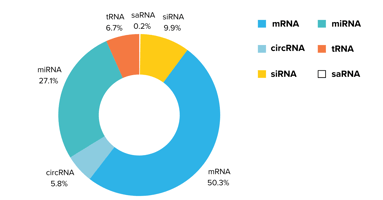
LNPs can deliver various RNA molecules, including mRNA, self-amplifying RNA (saRNA), circular RNA (circRNA), and microRNA (miRNA), as well as other nucleic acid-based medicines like plasmid DNA (pDNA).

Lipid nanoparticles' (LNP) payload options include mRNA, circRNA, miRNA, and pDNA among others.
Read more about RNA types that can be encapsulate
Advantages of LNPs-based delivery
LNPs offer distinct advantages over alternative delivery systems, making them the leading platform for RNA therapeutics:
-
Reduced Immunogenicity: Unlike viral vectors, LNPs minimize immune activation, improving safety for repeated dosing.
-
Scalable and Reproducible: LNP formulations can be rapidly scaled for clinical and commercial applications with consistent quality and low batch-to-batch variability.
-
Customizable Pharmacokinetics: PEG-lipids and lipid composition allow tunable circulation time, biodistribution, and tissue targeting.
LNP structure and composition
LNPs consist of four key lipid components:
-
Ionizable lipids: Facilitate RNA encapsulation and endosomal escape (e.g., MC3, SM-102, ALC-0315, C12-200).
-
Helper lipids: Enhance stability and membrane fusion (e.g., DSPC, DOPE).
-
Cholesterol: Improves membrane fluidity and nanoparticle integrity.
-
PEG-lipids: Modulate pharmacokinetics, reduce aggregation, and can be used to anchor ligands such as antibodies or peptides.
The interplay of these components dictates RNA encapsulation efficiency, stability, and biodistribution. Custom formulations allow the optimization of payload protection, controlled release, and targeted delivery.
Common challenges in RNA-LNP synthesis
1. RNA stability and degradation
Challenge:
RNA molecules are highly susceptible to degradation by ribonucleases (RNases) and can lose functionality if not properly protected. This instability can reduce the efficacy of RNA-based therapeutics.
Solution:
-
Increase encapsulation efficiency: Optimizing LNP composition ensures a high RNA encapsulation rate, shielding it from enzymatic degradation.
-
Modified nucleotides: Incorporating chemically modified nucleotides (e.g., pseudouridine in mRNA) enhances RNA stability by preventing degradation and reducing immunogenicity.
-
Advanced storage conditions: Optimized buffer formulations extend RNA-LNP shelf life.
Our RNA-LNP encapsulation process
At CATUG, we utilize our proprietary technology SizeSmart™ to control LNP's size and MaxMix™ to manufacture RNA-LNP products at small and large scales, from R&D to GMP.
Additionally, our MultiPlus™ technology allows for the encapsulation of multiple mRNAs within a single LNP. Encapsulate single and multiple mRNAs, pDNA, circular RNA, siRNA, saRNA, and sgRNA+mRNA. See all our products here.

CATUG's RNA-LNP manufacturing process includes proprietary technologies such as SizeSmart™, MaxMix™, and MultiPlus™ for large and small-scale manufacturing.
Our in-house manufacturing pipeline provides LNPs with minimal batch variability, precise size control (60-100 nm), and >95% encapsulation efficiency, maximizing outcomes and minimizing costs.
2. Immune response and reactogenicity
Challenge:
Unmodified RNA and LNP can activate innate immune sensors, such as toll-like receptors (TLRs), leading to inflammatory responses that reduce therapeutic efficacy and safety.
Solution:
-
PEGylation sweet spot: Achieving a fine balance of PEG-lipids is essential. While these lipids can improve circulation time and reduce immune recognition, they also hinder LNP uptake by target cells.
-
RNA engineering: Using modified nucleosides (e.g., 1-methylpseudouridine (Ψ), 5-Methylcytidine (m5C)) minimizes immune activation.
We have an extensive library of LNP formulations, screened and validated for optimal performance in your intended applications. Talk to our experts and find the best LNP solution for you.
3. Batch-to-batch consistency and scalability
Challenge:
Reproducibility in LNP formulation is critical for clinical and commercial applications. However, variations in lipid synthesis, mixing techniques, and nanoparticle assembly can lead to inconsistencies.
Solution:
-
Proprietary mixing technology: Our in-house methods ensure uniform LNP size and high encapsulation efficiency (>95%).
-
Automated GMP manufacturing: Our state-of-the-art GMP-compliant facilities use automated processes to minimize human error and ensure precise control over formulation parameters. This reduces batch-to-batch variability and facilitates scalability.
-
Deep analytical characterization: At CATUG, we have developed more than 70 analytical methods to perform extensive quality control analysis.
Our in-house analytical methods enable the study of 50+ RNA and LNP quality attributes, ensuring precise sample control and minimal batch-to-batch variability.
4. Biodistribution and targeted delivery
Challenge:
Unmodified LNPs often accumulate in the liver due to their natural tropism, limiting their use for non-hepatic targets. Additionally, after cellular uptake via endocytosis, LNPs often get trapped in endosomes, leading to inefficient release of RNA cargo.
Solution:
-
Targeted delivery strategies: Ligand-functionalized LNPs (e.g., aptamer- or antibody-conjugated LNPs) enhance uptake in specific cell types.
-
Charge and hydrophobicity optimization: Adjusting lipid ratios or modulating surface properties improves circulation time and tissue penetration.
-
Endosomal escape enhancers: Incorporating fusogenic lipids or endosome-disrupting peptides improves RNA release.
Our proprietary SizeSmart™ technology tunes LNP size according to your project needs and the desired route of administration.
Have a project in mind? Let's explore a formulation that fits your specific route of administration or LNP biodistribution.
5. Regulatory and manufacturing challenges
Challenge:
Scaling from research to GMP-compliant production presents regulatory hurdles, including stringent quality control and reproducibility requirements.
Solution:
-
GMP-compatible raw materials: We use high-purity lipids and enzymes to meet regulatory standards. Each compound undergoes rigorous screening and testing to ensure the highest quality raw materials.
-
Standardized Characterization Methods: Our rigorous testing ensures safety and efficacy.
-
Regulatory Documentation Support: Get CMC (Chemistry, Manufacturing, and Controls) documentation streamlined for regulatory approvals.
Our high-quality manufacturing delivers GMP-grade, IND-enabling products to support your project. From R&D to GMP, you get support at every production scale.
Our manufacturing capabilities: scalable, GMP-grade LNP production
- Preclinical formulation & process development
- Clinical-scale GMP manufacturing
- Analytical & quality control services
-
Preclinical formulation & process development
✔ High-throughput LNP formulation screening
✔ Custom RNA-LNP synthesis for specific therapeutic applications
✔ Experienced in gene editing, CAR-T therapy, mRNA vaccines, and other RNA therapeutics
-
Clinical-scale GMP manufacturing
✔ End-to-end GMP production of RNA-LNP.
✔ Scalable manufacturing for low batch-to-batch variation and high reproducibility
✔ Sterile filtration and aseptic fill-finish services
-
Analytical & quality control services
✔ Encapsulation efficiency (>95%) and RNA integrity analysis
✔ Particle size (DLS, NTA) and ζ-potential measurements
✔ Residual impurities and endotoxin testing for regulatory compliance
We provide custom LNP formulation consultation, supporting your RNA therapeutics from synthesis to commercialization.